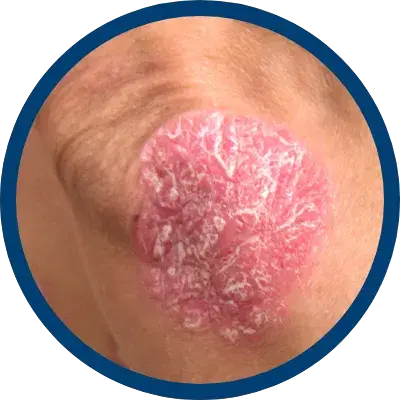
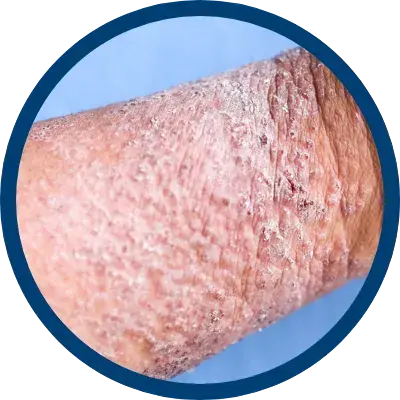
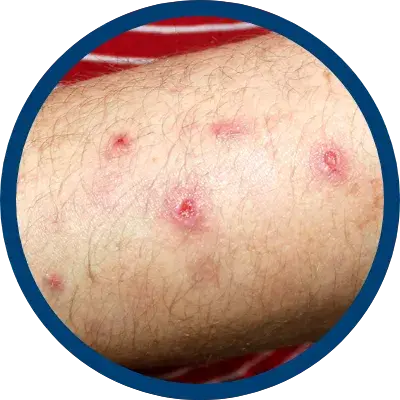
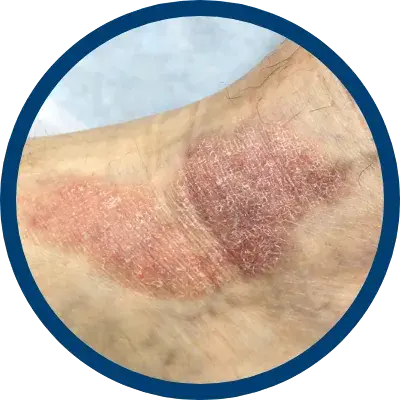
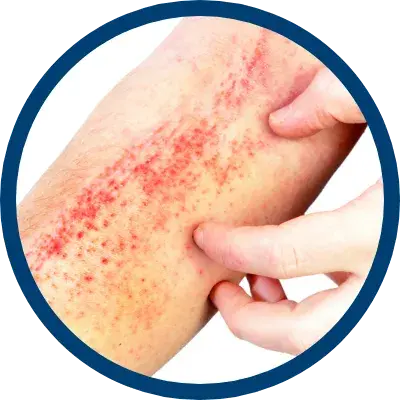
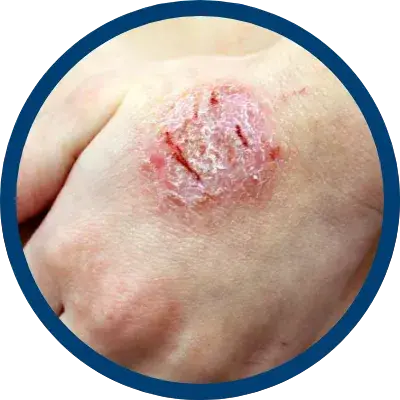
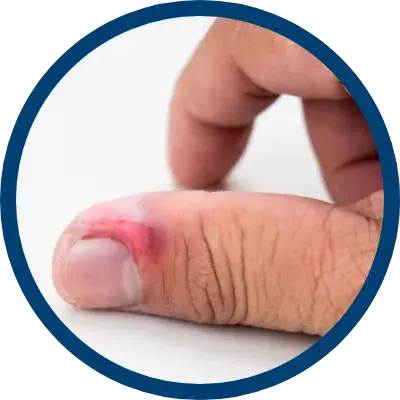
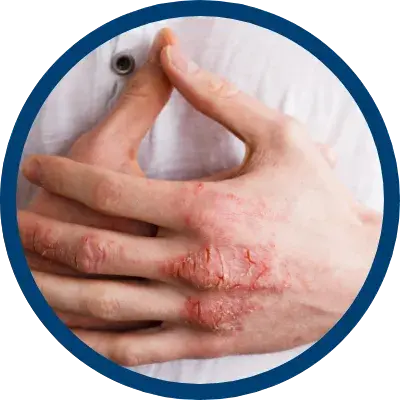
INDICATIONS AND USAGE
CORDRAN®Tape (Flurandrenolide Tape, USP) is a corticosteroid
indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid
responsive dermatoses, particularly dry, scaling localized lesions.
IMPORTANT SAFETY INFORMATION
Topical corticosteroids are contraindicated in patients with a history of
hypersensitivity to any of the components of these preparations. Use of
CORDRAN®Tape is not recommended for lesions exuding serum or in intertriginous
areas.
Systemic absorption of topical corticosteroids has produced reversible
hypothalamic-pituitary-adrenal (HPA) axis suppression, manifestations of Cushing’s syndrome,
hyperglycemia, and glucosuria in some patients. Use over large surface areas, prolonged use,
and the addition of occlusive dressings augment systemic absorption. Pediatric patients may
absorb proportionately larger amounts of topical corticosteroids and thus may be more
susceptible to systemic toxicity.
HPA axis suppression, Cushing’s syndrome, and intracranial hypertension have
been reported in pediatric patients receiving topical corticosteroids. Patients receiving a
large dose applied to a large surface area should be evaluated periodically for evidence of
HPA axis suppression, and therapy should be modified or discontinued as appropriate.
Topical corticosteroids should be used during pregnancy only if the potential
benefit justifies the potential risk to the fetus. Drugs of this class should not be used
extensively for pregnant patients or in large amounts or for prolonged periods of time.
Caution should be exercised when topical corticosteroids are administered to a nursing
woman.
Local adverse reactions may occur more frequently with the use of occlusive dressings. These
reactions are listed in approximate decreasing order of occurrence: burning, itching,
irritation, dryness, folliculitis, hypertrichosis, acneiform eruptions, hypopigmentation,
perioral dermatitis, allergic contact dermatitis. Reactions that may occur more frequently
with occlusive dressings include: maceration of the skin, secondary infection, skin atrophy,
striae, and miliaria.
For more information on CORDRAN®Tape, please see
accompanying Full Prescribing Information.
To report an adverse event or product complaint, call or email:
Medical
Affairs and Customer Relations
Phone: 1-866-665-2782
Fax: 510-595-8183
Email: almirallmc@eversana.com
REFERENCES:
- US Department of Health and Human Services. Orange Book: Approved Drug Products with
Therapeutic Equivalence Evaluations. 39th ed. US Food and Drug Administration;
2019. https://www.fda.gov/drugs/informationondrugs/ucm129662.htm. Accessed April 9,
2019.
- Ference JD, Last AR. Choosing topical corticosteroids. Am Fam Physician.
2009;79(2):135-140.
- CORDRAN Tape [package insert]. Exton, PA: Almirall, LLC, 2018.
- Weiner MA. Flurandrenolone tape. A new preparation for occlusive therapy. J Invest
Dermatol. 1966;47(1):63-66.
- Medscape. Latest Medical News, Clinical Trials, Guidelines – Today on Medscape.
Medscape.com. https://www.medscape.com. Accessed May 16, 2019.
- Mullins TB, Sharma P, Sonthalia S. Prurigo Nodularis. Treasure Island, FL:
StatPearls Publishing; 2019.
- Charifa A, Badri T. Lichen Simplex Chronicus. Treasure Island, FL: StatPearls
Publishing; 2019.
- Fonacier L, Bernstein DI, Pacheco K, et al. Contact dermatitis: a practice
parameter-update 2015. J Allergy Clin Immunol Pract. 2015;3(3 Suppl):S1-39.
- Pongdee T. Contact Dermatitis Overview. American Academy of Allergy Asthma &
Immunology.
https://www.aaaai.org/conditions-and-treatments/library/allergy-library/contact-dermatitis.
Accessed June 25, 2019.
- Leggit JC. Acute and chronic paronychia. Am Fam Physician. 2017;96(1):44-51.
- Mayeaux EJ Jr. Paronychia. In: Usatine RP, Smith MA, Mayeaux EJ Jr., Chumley HS, eds.
The Color Atlas and Synopsis of Family Medicine. 3rd ed. New York, NY:
McGraw-Hill; 2018:1287-1291.
- Hand Eczema. National Eczema Association.
https://nationaleczema.org/eczema/types-of-eczema/hand-eczema. Accessed May 30, 2019.
- Abyaneh MAY, Griffith R, Falto-Aizpura L, Nouri K. Cryosurgery for psoriasis. In:
Abramovits W, Graham G, Har-Shai Y, Strumia R, eds. Dermatological Cryosurgery and
Cryotherapy. London, UK: Springer Verlag; 2016.
https://www.springer.com/us/book/9781447167648.